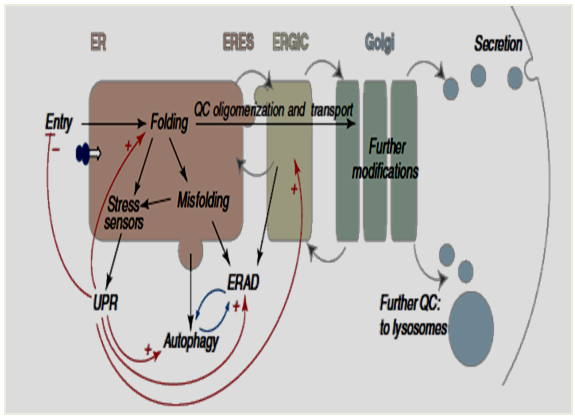
4 6 Given the substantial sequence identity between CNX and CRT it was not long before CRT was demonstrated to share with CNX the ability to bind transiently to diverse nascent proteins. 3 Since then, CNX has been shown to interact transiently with a wide array of newly synthesized membrane or soluble proteins that pass though the ER. Of T cell receptors and membrane immunoglobulins, 2 and also as a microsomal membrane protein that can be phosphorylated in vitro. Interacts with partially assembled class I histocompatibility molecules, 1 with partial complexes In 1991, CNX was discovered virtually simultaneously by three groups as a protein that The controversial issue of whether CNX/CRT function solely as lectins or also as “classical”Ĭhaperones that recognize the unfolded polypeptide portion of glycoproteins is presentedĪnd the evidence supporting current models is discussed in detail. Recent determination of the structure of the ER luminal domain of CNX and a portion ofĬRT. Organization and locations of functional sites have been revealed through mutagenesis and the Site, expression of CNX/CRT and model substrates in heterologous systems, gene disruption,Īnd reconstitution of function with purified components in vitro. Which include interfering with the recognition of glycoproteins through CNX/CRT's lectin Insights into how this function is accomplished have been provided through diverse approaches In this chapter we present the evidence that calnexin (CNX) and calreticulin (CRT) functionĪs molecular chaperones to assist in the folding and subunit assembly of the majority ofĪsn-linked glycoproteins that pass through the endoplasmic reticulum.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
